 
The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. The atomic number of each element increases by one, reading from left to right. 
Period A horizontal row in the periodic table. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in C2H5Cl: Molar Mass (g/mol) C (Carbon) 2 × 12.0107 24.0214. For example, if we are trying calculate for ammonia (NH 3 ), then we. The atomic mass is equal to the atomic number which is listed below the element symbol. Element Symbol Atomic weight Atoms Mass percent Phosphorus: P: 30.973762: 1: 100. Molar Mass of Ammonia NH 3 Step 1: The first step for calculating molar mass is to identify all the elements in a given molecule and write their atomic masses using the periodic table. Compound Moles Weight, g P: Elemental composition of P. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in HNO3: Molar Mass (g/mol) H (Hydrogen) 1 × 1.00794 1.00794. Members of a group typically have similar properties and electron configurations in their outer shell. Enter a chemical formula to calculate its molar mass and elemental composition: Molar mass of P (Phosphorus) is 30.9737620 ± 0.0000020 g/mol. It is similar to other counting units, such as a pair (2) and a dozen (12). Mathematically, the defining equation of molar mass is. 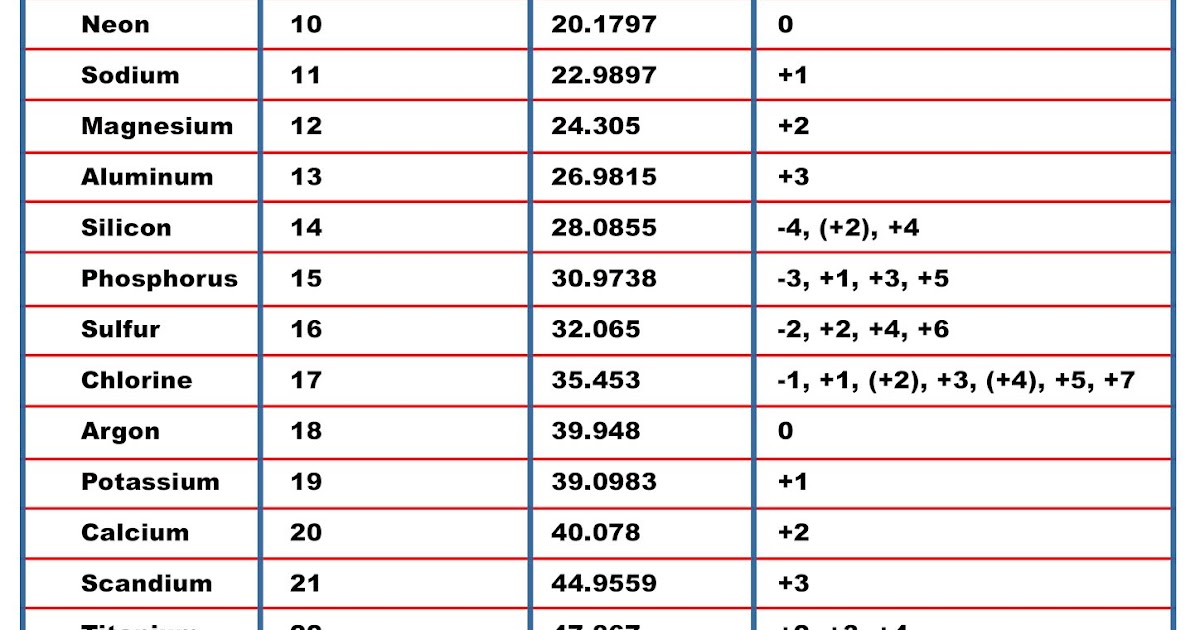
To find the mole fraction and percentage of each element in C3H8, divide each total from step 3 by the total molar mass found in step 4: Mole Fraction. The units of molar mass follow its definition grams per mole. 
Molar Mass Formula: A mole is a counting unit used to determine the number of molecules, atoms, ions, or molecular formula units in a certain compound. Finally, add together the total mass of each element to get the molar mass of C3H8: 36.0321 g/mol + 8.06352 g/mol 44.09562 g/mol. For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. Periodic Table Elements: Enter Molecular Formula. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
